Can you imagine fully charging your cell phone in just a few seconds?
Researchers in Drexel University’s College of Engineering can, and they took a big step toward making it a reality with their recent work unveiling of a new battery electrode design in the journal Nature Energy.
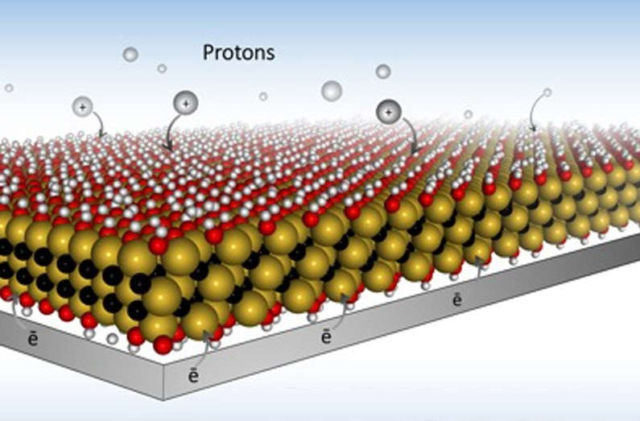
Using MXene Drexel they developed electrode designs, that allow for much faster charging, because they open up paths for ions to quickly travel within the material.
The team, led by Yury Gogotsi, PhD, Distinguished University and Bach professor in Drexel’s College of Engineering, in the Department of Materials Science and Engineering, created the new electrode designs from a highly conductive, two-dimensional material called MXene.
Their design could make energy storage devices like batteries, viewed as the plodding tanker truck of energy storage technology, just as fast as the speedy supercapacitors that are used to provide energy in a pinch — often as a battery back-up or to provide quick bursts of energy for things like camera flashes.
Gogotsi said:
“This paper refutes the widely accepted dogma that chemical charge storage, used in batteries and pseudocapacitors, is always much slower than physical storage used in electrical double-layer capacitors, also known as supercapacitors. We demonstrate charging of thin MXene electrodes in tens of milliseconds. This is enabled by very high electronic conductivity of MXene. This paves the way to development of ultrafast energy storage devices than can be charged and discharged within seconds, but store much more energy than conventional supercapacitors.”
via sciencealert




Leave A Comment