Scientists developed a system that produces electricity and hydrogen (H2) while eliminating carbon dioxide (CO2).
The recent study affiliated with UNIST, turned carbon emissions, which is the main contributor of global warming, into electricity and hydrogen.
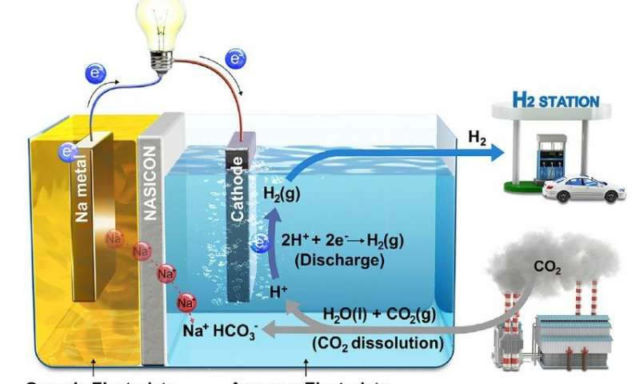
In this work, led by Professor Guntae Kim in the School of Energy and Chemical Engineering at UNIST, the research team presented Hybrid Na-CO2 system that can continuously produce electrical energy and hydrogen through efficient CO2 conversion with stable operation for over 1,000 hr from spontaneous CO2 dissolution in aqueous solution.
Above, schematic illustration of Hybrid Na-CO2 System and its reaction mechanism. Credit UNIST
Professor Guntae Kim, explains:
“Carbon capture, utilization, and sequestration (CCUS) technologies have recently received a great deal of attention for providing a pathway in dealing with global climate change. The key to that technology is the easy conversion of chemically stable CO2 molecules to other materials. Our new system has solved this problem with CO2 dissolution mechanism.”
source UNIST





Leave A Comment